|
Further, we observed that the infinite nitrogen chain and the N 6 ring network structure contain typical covalent bonds formed by the hybridization of the sp 2 and sp 3 orbitals of N, respectively. 
We found that SrN 6 has a new P phase at a pressure of 22 GPa and an infinite nitrogen chain structure, and BaN 6 has a new C2/ m phase at 110 GPa, with an N 6 ring network structure. In order to find other stable crystal structures, we have performed XN 6 (X = Ca, Sr and Ba) exploration under high pressure. The N-rich compounds XN 6 (X = Ca, Sr and Ba) have been reported under normal pressure. In recent years, to explore N-rich compounds which are stable and easy to synthesize has become a new research direction. Nitrogen can exist in alkaline earth metal nitrogen-rich (N-rich) compounds in the form of single or double bonds. These levels may be higher at hazardous waste sites.Multi-nitrogen or polynitrogen compounds can be used as potential high energy-density materials, so they have attracted great attention. Barium is found in most land soils at low levels. 
Barium compounds that are persistent usually remain in soil surfaces, or in the sediment of water soils. When fish and other aquatic organisms absorb the barium compounds, barium will accumulate in their bodies.īecause it forms insoluble salts with other common components of the environment, such as carbonate and sulphate, barium is not mobile and poses little risk. Because of their water-solubility these barium compounds can spread over great distances. Some barium compounds that are released during industrial processes dissolve easily in water and are found in lakes, rivers, and streams. There is no proof that barium can cause infertility or birth defects. Small amounts of water-soluble barium may cause a person to experience breathing difficulties, increased blood pressures, heart rhythm changes, stomach irritation, muscle weakness, changes in nerve reflexes, swelling of brains and liver, kidney and heart damage.īarium has not shown to cause cancer with humans. The uptake of very large amounts of barium that are water-soluble may cause paralyses and in some cases even death. Barium compounds that dissolve in water can be harmful to human health. The health effects of barium depend upon the water-solubility of the compounds. The exposure will than be caused by breathing dust, eating soil or plants, or drinking water that is polluted with barium. People that live near them may be exposed to harmful levels. Many hazardous waste sites contain certain amounts of barium. Most of the health risks that they can undergo are caused by breathing in air that contains barium sulphate or barium carbonate. People with the greatest risk to barium exposure with additional health effects are those that work in the barium industry. The amount of barium that is detected in food and water usually is not high enough to become a health concern. Each year about 6 million tonnes are produced and reserves are expected to exceed 400 million tonnes. The main mining areas are UK, Italy, Czech Republic, USA and Germany. The chief mined ores are barite, which is also the most common and witserite. It can also enter the air during coal and oil combustion. As a result barium concentrations in air, water and soil may be higher than naturally occurring concentrations on many locations.īarium enters the air during mining processes, refining processes, and during the production of barium compounds. High amounts of barium may only be found in soils and in food, such as nuts, seaweed, fish and certain plants.īecause of the extensive use of barium in the industries human activities add greatly to the release of barium in the environment. 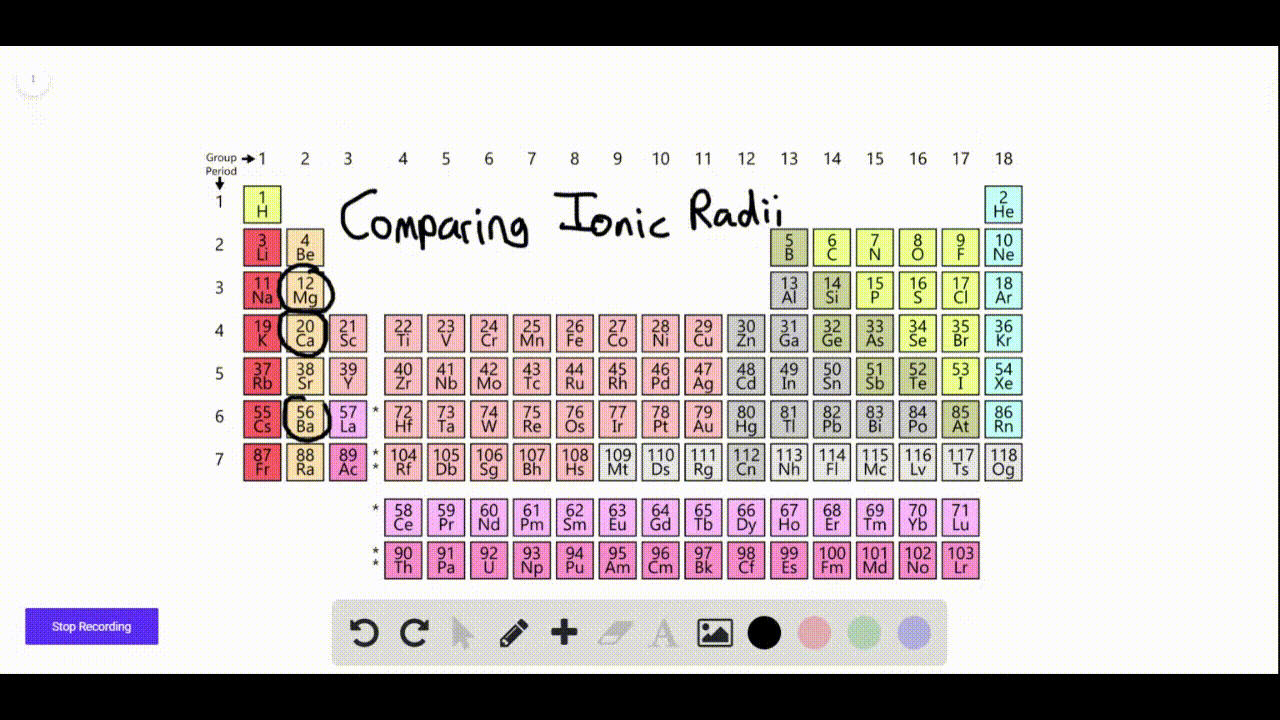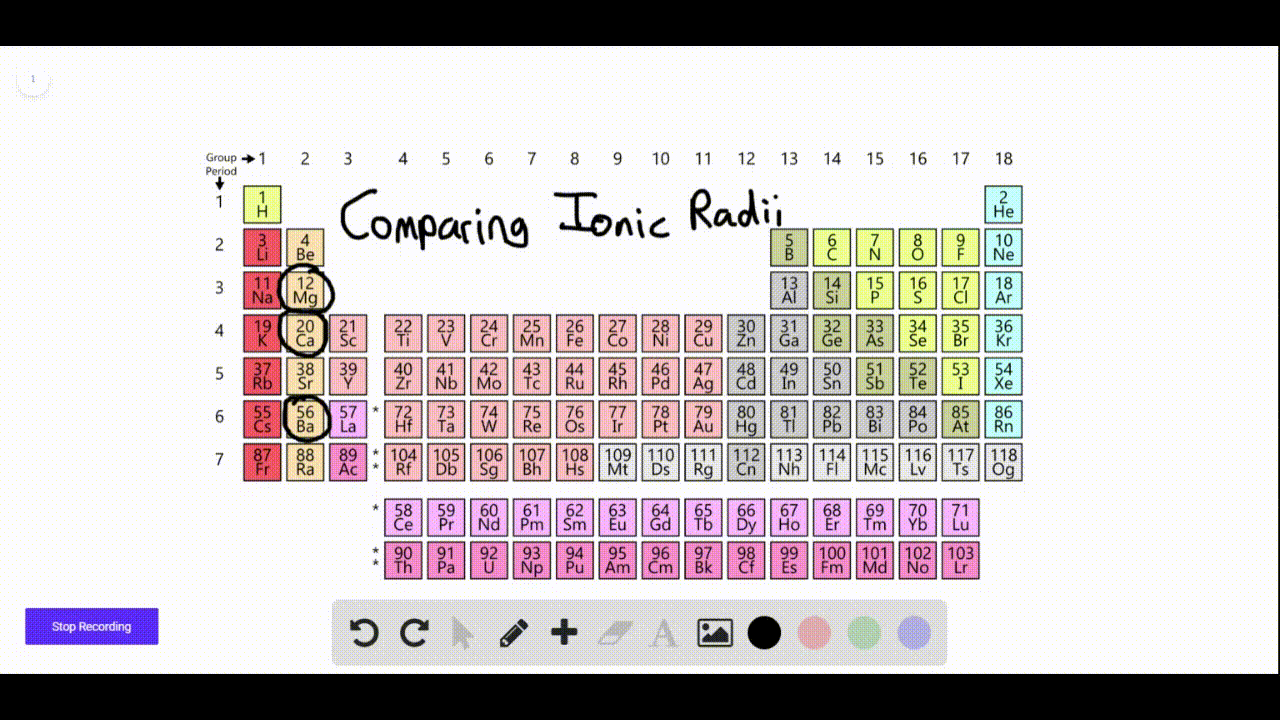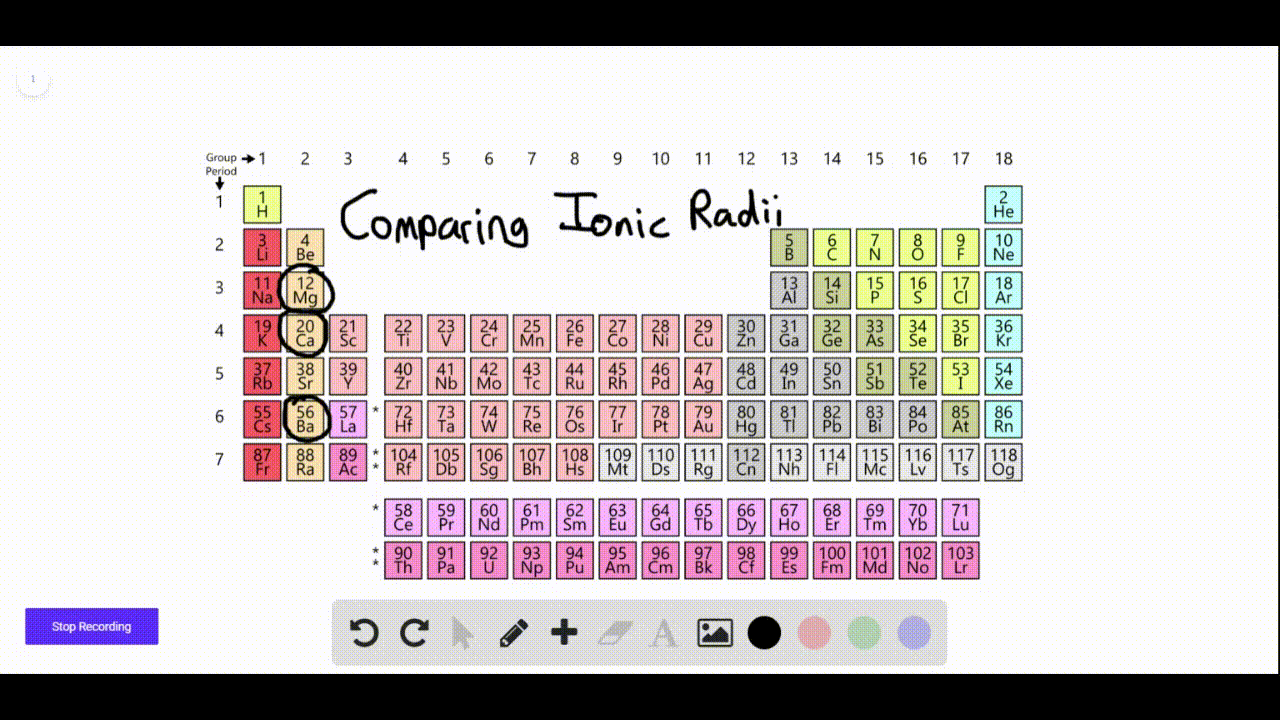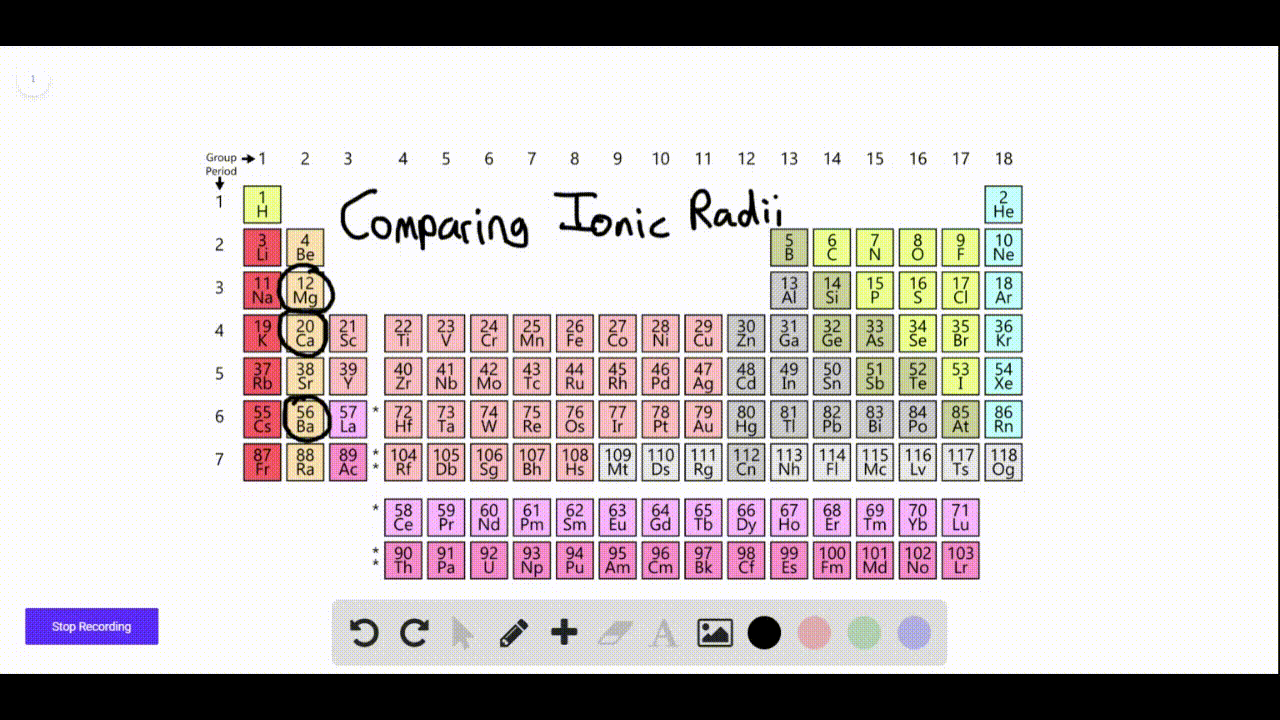
Barium nitrate and clorate give fireworks a green colour.īarium is surprisingly abundant in the Earth's crust, being the 14th most abundant element. Drilling mud simplifies drilling through rocks by lubricating the drill.īarium compounds are also used to make paint, bricks, tiles, glass, and rubber. 
It is also used in fluorescent lamps: impure barium sulfide phosphoresces after exposure to the light.īarium compounds are used by the oil and gas industries to make drilling mud. Barium reacts with almost all the non-metals, forming often poisouning compounds.īarium is often used in barium-nickel alloys for spark-plug electrodes an in vacuum tubes as drying and oxygen-removing agent. Barium oxidizes in air, reacts vigoroulsy with water to form the hydroxide, liberating hydrogen. Ii is very light and its density is half that of iron. It occurs combined with other chemicals, such as sulfur, carbon or oxygen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
